
Improperly ventilated kerosene lanterns and stoves can generate dangerously high carbon monoxide levels in enclosed spaces. If there is too little oxygen, or too much carbon, present when something burns, the burning produces carbon monoxide (CO) instead of (or as well as) carbon dioxide (CO 2). This means that someone who breathes too much CO can have their hemoglobin become saturated with it, making it impossible for the blood to deliver oxygen to their cells.Ĭarbon monoxide is often a product of incomplete combustion. Hemoglobin, the protein in blood that carries oxygen from the lungs to cells throughout the body, is more than 200 times more prone to bind with carbon monoxide than it is with oxygen. However, the gas is quite poisonous to humans and other air-breathing creatures that need oxygen. Scientists estimate than the total annual production of carbon monoxide, from human and natural sources, is between two and five gigatons.Ĭarbon monoxide is a relatively unreactive compound, so it doesn't pose much threat to plants or exposed materials. In the United States, about 2/3rds of carbon monoxide emissions come from automobiles. Certain industrial processes, along with fossil fuel and biomass burning, are major human-produced sources of CO. In areas with heavy traffic, CO levels can rise to as high as 50 ppm. Atmospheric carbon monoxide levels in typical urban areas are around 10 ppm (parts per million), about 100 times higher than in Earth's atmosphere overall. Carbon monoxide is the most abundant, by mass, pollutant gas. Humans contribute vast quantities of CO to our atmosphere, mostly as a result of automobile emissions. Volcanic gases contain between 0.01 and 2% carbon monoxide. Natural sources of carbon monoxide in Earth's atmosphere include volcanoes and bushfires. The gas eventually reacts with oxygen (O 2) to form carbon dioxide (CO 2). 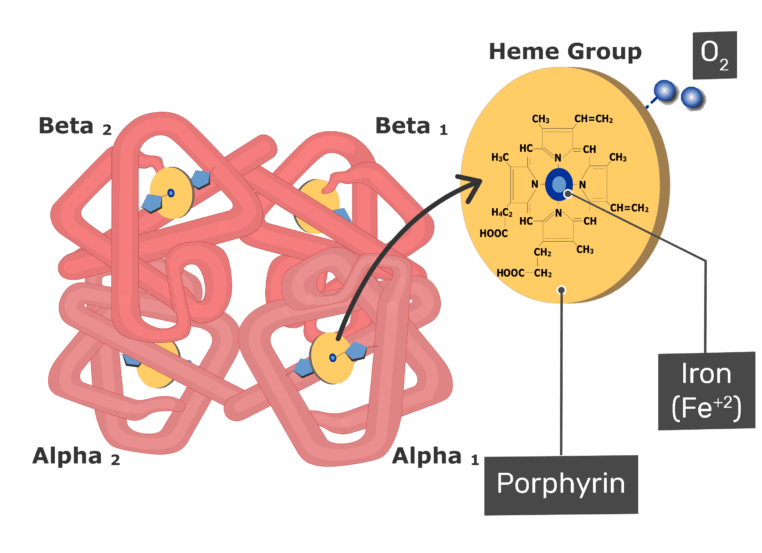
Carbon monoxide has a typical "lifespan" of several months in Earth's atmosphere. A typical concentration of CO in Earth's troposphere is around 100 ppb (parts per billion meaning one hundred out of every billion air molecules is carbon monoxide), although especially clean air can have concentrations as low as 50 ppb. A molecule of carbon monoxide (CO) contains one carbon atom and one oxygen atom.Ĭarbon monoxide is present in Earth's atmosphere at very low concentrations. It is also flammable and is quite toxic to humans and other oxygen-breathing organisms. In the models, the carbon atom is gray and the oxygen atom is red.Ĭarbon monoxide is a colorless, odorless, tasteless gas. This illustration shows four different ways that chemists represent carbon monoxide.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
